Learning Target for Week 4
By the end of this week, I will be able to explain what forces act on matter and why atoms are related to biology by completing sections below to summarize teacher lecture notes, textbook notes and individual question analysis. Italicized notes are to help and should not be included in student portfolios.
The Chemistry of Life (Chapter 2: Background for Biochemistry)
A. Key Forces Acting on Matter
|
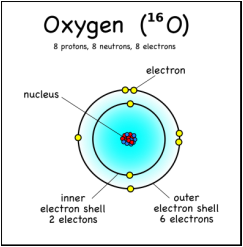
C. The AtomThe Quantum Mechanical Model of the atom is the theory of the atom that says that all the properties that can be measured of a particle can be calculated from its orbits. People thought that an atom was a black circle.Scientists experimented and found out that there's energy between the orbits "dark matter" something else that was unknown but is present in the atom.Also they found e-clouds and that the neutrons and protons are hold together in the nucleus. |
B. Matter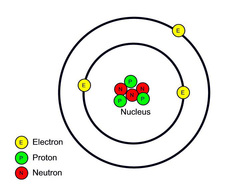
1. Atoms: a basic unit of matter with a specific number of protons for each element. Atoms have a zero charge when they're alone.This means that the number of electrons will equal the number of protons in an atom.
source:Atom> 
2. Elements: the form of matter in which the atoms are similar.
Source: element> 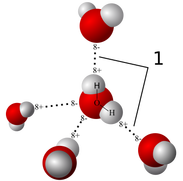
3. Compounds: 2 or more elements chemically bonded.
Source:compound> 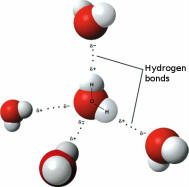
4. Molecules: 2 or more atoms chemically bonded.
Source:molecule> 
5. Ions: an atom that has gained electric charge by losing or getting one or more electrons, is charged due to a different number of protons and electrons causing a charged atom.
Source:ion> 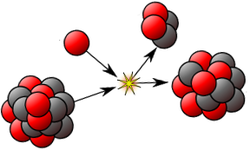
6. Radioactivity:energy released when atomic nuclei are unstable due to the ratio of protons to neutrons.
Source:radioactivity> 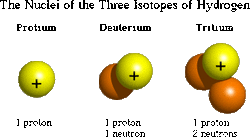
7. Isotopes:an element with a different number of neutrons, but with the same number of protons.
Source:isotope> E. Chemical Bonds
|
F. Water and It's Importance to Lifeinclude the following in your discussion about water:
|